The perils of PCR-based diagnosis of Clostridioides difficile infections: Painful lessons from clinical trials.
Authors of this article are:
Kong LY, Davies K, Wilcox MH.
A summary of the article is shown below:
Diagnostic tests favoured to detect C. difficile infections (CDI) have undergone successive changes. The problem of over-diagnosis with polymerase chain reaction (PCR) testing is recognized in the clinical setting; here we discuss the parallel of the clinical trial setting. We summarize and discuss four examples of the impact of method used to diagnose CDI on clinical trial outcomes. Bezlotoxumab, a human monoclonal antibody neutralizing toxin B, was found to be protective against recurrent CDI (rCDI) in clinical trials. A post hoc analysis showed that the magnitude of the relative reduction in rCDI rates of bezlotoxumab over placebo in patients diagnosed with toxin-based testing was almost double that in patients diagnosed with PCR. SER-109, a microbiome therapeutic developed to prevent rCDI, showed promise in a phase 1b trial, but results were not replicated in a phase 2 trial in which diagnosis was in majority PCR-based. Surotomycin, an oral lipopeptide antibiotic, was found to be non-inferior to vancomycin in phase 2 study, but development was discontinued after unfavourable phase 3 results in which the majority of CDI were diagnosed by PCR. Finally, a C. difficile vaccine program for a toxoid vaccine developed by Sanofi/Pasteur was terminated after interim analysis of a phase 3 trial, in which CDI diagnosis was based solely on PCR. We highlighted the perils of using PCR alone in studies involving different aspects of C. difficile clinical research, including immunotherapies, microbiome-based therapies, treatments, and vaccines. The importance of designing C. difficile clinical trials with careful consideration to the diagnostic testing method cannot be overemphasized.Copyright © 2019 Elsevier Ltd. All rights reserved.
Check out the article’s website on Pubmed for more information:
[link-preview url=https://www.ncbi.nlm.nih.gov/pubmed/31201853 forceshot=true]
This article is a good source of information and a good way to become familiar with topics such as: Clinical trials; Clostridioides difficile; Diagnosis.
NativeFolder: The Only Bacterial Culture Medium for the Expression of Soluble Proteins
-
- Sale!
- Molecular Biology, Research Kits
NativeFolder Bacterial Culture Medium
- Original price was: $795.00.$295.00Current price is: $295.00.
Interference Test Kit for Assay Validation
-
- Sale!
- Interference, Research Kits
Interference Test Kit
- Original price was: $995.00.$495.00Current price is: $495.00.
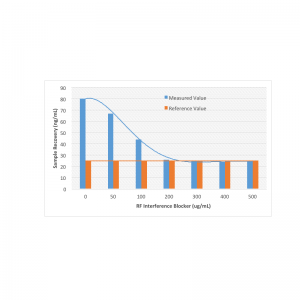
Rheumatoid Factor Interference Blocker
-
- Sale!
- Antibodies, Interference
Rheumatoid Factor Interference Blocker
- Original price was: $595.00.$395.00Current price is: $395.00.
New Antibodies from MOLECULAR DEPOT
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CTNL1 CTNNAL1 Antibody (Human, Mouse)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Monoclonal CXCL11 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal Cullin 4a (CUL4A) Antibody (Human)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal Cullin 1/CUL1 (N-Terminus) Antibody (Human, Mouse, Rat)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CTNA2 CTNNA2 Antibody (Human, Mouse)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Monoclonal CTR1/SLC31A1 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Mouse Monoclonal CTLA4 Antibody
- Original price was: $1,795.00.$895.00Current price is: $895.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CXCL5 (A38-Q130) Antibody (Rat)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CXCR1 (N-Terminus) Antibody (Human)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Anti-CXB5 GJB5 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
New Proteins from MOLECULAR DEPOT
-
- Sale!
- Conjugates, Proteins
Captopril HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Aminopenicillic Acid HRP Conjugate
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Biochemistry, Proteins
PKC-α, Active, GST-tagged from Xanopus sp.
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Conjugates, Proteins
Prilocain HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
T3 (Triiodothyronine) HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Pipemidic acid HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Biochemistry, Proteins
PKD2 Protein, Active (Recombinant Human)
- Original price was: $1,795.00.$895.00Current price is: $895.00.
-
- Sale!
- Conjugates, Proteins
Meropenem BSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Piperacillin HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Biochemistry, Proteins
Protein A Peroxidase from Staphylococcus aureus/horseradish
- Original price was: $1,195.00.$595.00Current price is: $595.00.
New Chemicals from MOLECULAR DEPOT
-
- Sale!
- Chemicals, Interference, Lipids
Triglyceride Mix for Interference Testing
- Original price was: $895.00.$395.00Current price is: $395.00.
-
- Sale!
- Chemicals
Microparticle Stabilizer Solution
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Enzyme Acceptor Stabilization Buffer
- Original price was: $495.00.$250.00Current price is: $250.00.
-
- Sale!
- Buffers & Solutions, Chemicals
Enzyme Donor Stabilization Buffer
- Original price was: $495.00.$250.00Current price is: $250.00.
-
- Sale!
- Chemicals, Conjugates
Progesterone Biotin Conjugate Solution
- Original price was: $650.00.$350.00Current price is: $350.00.
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Activation Buffer
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Washing Buffer
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Blocking Buffer
- Price range: $395.00 through $695.00
-
- Sale!
- Buffers & Solutions, Chemicals
Latex Microparticles Anti Aggregation Agent
- Price range: $295.00 through $3,500.00
-
- Sale!
- Chemicals
CPRG (Chlorophenol red-β-D-galactopyranoside)
- Original price was: $850.00.$395.00Current price is: $395.00.