Beta-galactosidase Enzyme Donor
Original price was: $795.00.$450.00Current price is: $450.00.
Catalog #: P2010004 (250 μg)
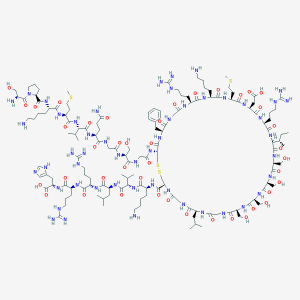
Beta-galactosidase Enzyme Donor (250 ug) consists of the Alpha Peptide fragment of the enzyme can Beta-galactosidase. This product can complement the Beta-galactosidase Enzyme Acceptor (also known as Omega domain) to reconstitute the fully active Beta-galactosidase enzyme. This phenomenon, known as alpha complementation, can be used to develop detection and tracing methods for various small molecules. The sequence of the enzyme donor has been specifically engineered to include a unique central cysteine that can be used for analyte conjugation. This product has a molecular weight of 11 kDa (99 amino acids) and is supplied as a white lyophilized powder. Custom bulk orders of this product are available upon request.
Live Enquiry about this product via Text/SMS: 1-858-900-3210 (8 am – 8 pm PST)
In stock
Product Description
Beta-galactosidase Enzyme Donor (alpha peptide)
| Catalog # | P2010004 |
| Size | 250 μg |
| Other Names | Beta-galactosidase Alpha Peptide |
| Supplied as | White lyophilized powder. |
| Molecular Weight | 11 kDa (99 amino acids) |
| Purity | >95% (SDS PAGE) |
| Storage | -20°C. Avoid repeated freeze/thaw cycles. |
| Suggested buffer | Beta-galactosidase Enzyme-Donor Stabilization Buffer (B2010001) |
| Keywords | Beta-galactosidase Alpha Peptide, Enzyme Donor, alpha complementation, LacZ. |
| Related products | Beta-galactosidase Enzyme Acceptor (Omega Domain), Beta-galactosidase Enzyme-Donor Stabilization Buffer, Beta-galactosidase Alpha Complementation Kit. |
Download Alpha Complementation Product Line Brochure.
About Beta-galactosidase Alpha Complementation
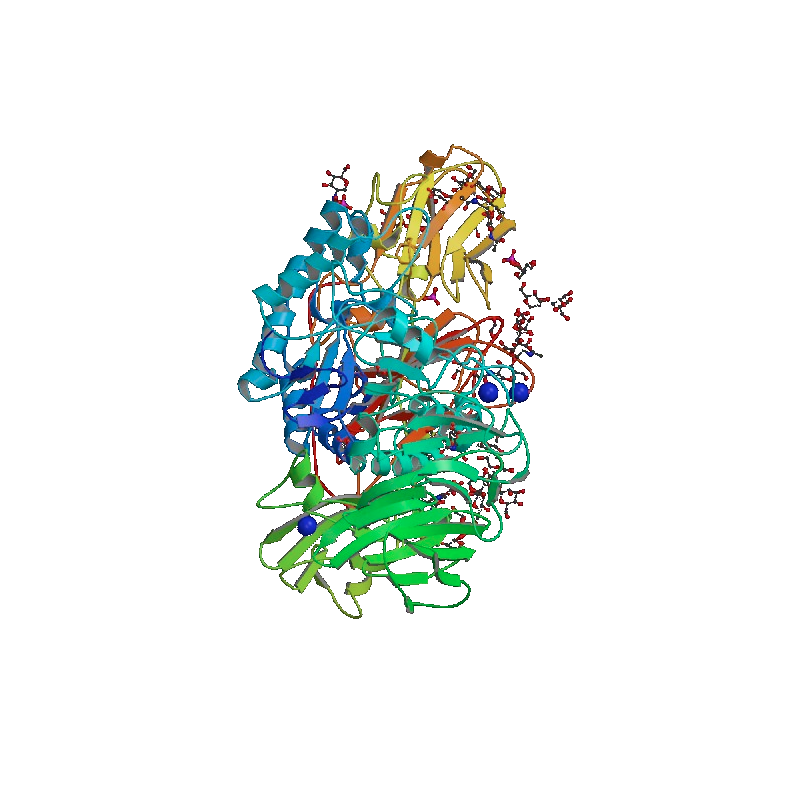
Beta-galactosidase Alpha-Complementation is a biochemical phenomenon first documented by Agnes Ullmann, while working in the lab of François Jacob and Jacques Monod. By means of molecular cloning, the native E. coli β-galactosidase enzyme can be split in two inactive fragments of different sizes. The smaller fragment, known as the alpha-peptide or enzyme donor, is about 100 amino residues in length and is inactive on its own (incapable of hydrolyzing a β-galactosidase substrate). The larger fragment, known as the omega fragment or enzyme acceptor, is about 900 amino residues in length and is also inactive on its own. Upon mixing the enzyme donor with the enzyme acceptor, the β-galactosidase enzyme is reconstituted and is now capable of hydrolyzing colorimetric substrates such as ONPG.
Both enzyme donor and enzyme acceptor can be cloned and expressed in special E. coli strains to yield highly pure, zero-background enzyme fragments (i.e. an enzyme donor and enzyme acceptor without measurable catalytic activities, when assayed individually). Interestingly, it was discovered that various analytes can be conjugated to the enzyme donor moiety and the enzyme donor-enzyme acceptor association modulated by an analyte-binding molecule (such as an antibody). As a result, an alpha-complementation-based assay can be developed.
References
- Kras, E. (2019). Beta-galactosidase: properties, structure and functions. New York: Nova Science Publishers.
- Arndt, T. (2017). Cloned Enzyme Donor Immunoassay. Lexikon Der Medizinischen Laboratoriumsdiagnostik, 1–2.
- Jeon, S. I., Yang, X., and Andrade, J. D. (2004). Modeling of homogeneous cloned enzyme donor immunoassay. Analytical Biochemistry, 333(1), 136–147.
- Tachi, T., Kaji, N., Tokeshi, M., and Baba, Y. (2009). Microchip-based Homogeneous Immunoassay Using a Cloned Enzyme Donor. Analytical Sciences, 25(2), 149–151.
- Khanna, P. L., and Worthy, T. E. (1993). CEDIA: A Recombinant-Based Homogeneous Enzyme Immunoassay.
Related Products
Additional Information
| Weight | 0.15 oz |
|---|---|
| Dimensions | 2 × 0.5 × 0.5 in |
Several ways to order Molecular Depot products:
1- Order online using our website’s automated shopping cart and checkout system (available 24/7).
2- Order by email: info@moleculardepot.com (we accept electronic PO, available 24/7).
3- Order by Live Chat.
4- Order by Fax: 1-858-437-9884 (available 24/7).