Evaluation of the Seegene Allplex™ Respiratory Panel for diagnosis of acute respiratory tract infections.
Authors of this article are:
Vandendriessche S, Padalko E, Wollants E, Verfaillie C, Verhasselt B, Coorevits L.
A summary of the article is shown below:
Objectives: The Seegene AllplexTM Respiratory panel was retrospectively challenged using a collection of quality control samples (QCMD) and clinical samples previously analysed with validated routine methods. Methods: A collection of 111 samples [43 QCMD samples, 13 bronchoalveolar lavage fluids and 55 nasopharyngeal aspirates/swabs] was tested with Seegene AllplexTM. The clinical samples were tested previously using either FTD® Respiratory Pathogens 21 qPCR assay (Fast Track Diagnostics), an in-house multiplex PCR for Bordetella, or BioGX Sample-ReadyTM Atypical pneumo panel (Becton Dickinson). Samples were stored at -80°C prior to analysis with Seegene Allplex™, nucleic acids were automatically extracted with NucliSENS Easymag (bioMérieux). Samples returning discordant results were subjected to repeat testing and/or additional testing by reference laboratories. Results: Seegene correctly identified 41/43 QCMD samples (95.4%); two samples positive for respiratory syncytial virus (RSV) and human metapneumovirus, respectively, were only correctly identified following repeat testing. In the 56 clinical samples, overall, 97 pathogens were identified: 65 pathogens (67.0%) were detected both by routine methods and Seegene, 24 pathogens (24.7%) only by routine methods, and 8 pathogens (8.2%) only by Seegene. The majority of discordant results was detected in samples with low pathogen load (22/32, 68.8%) and in samples containing multiple pathogens (25/32, 78.1%). Full agreement between methods was observed for influenza, RSV, adenovirus, Bordetella (para)pertussis and Chlamydia pneumoniae. Discordance was observed for human metapneumovirus, coronavirus OC43, bocavirus and parainfluenza virus, mainly type 4. Conclusion: Overall, the Seegene AllplexTM assay performed well for routine detection of important respiratory targets. Acceptable agreement was observed between Seegene and other routine assays.
Check out the article’s website on Pubmed for more information:
[link-preview url=https://www.ncbi.nlm.nih.gov/pubmed/30307378 forceshot=true]
This article is a good source of information and a good way to become familiar with topics such as: Respiratory pathogens; molecular detection; multiplex PCR; real-time PCR.
NativeFolder: The Only Bacterial Culture Medium for the Expression of Soluble Proteins
-
- Sale!
- Molecular Biology, Research Kits
NativeFolder Bacterial Culture Medium
- Original price was: $795.00.$295.00Current price is: $295.00.
Interference Test Kit for Assay Validation
-
- Sale!
- Interference, Research Kits
Interference Test Kit
- Original price was: $995.00.$495.00Current price is: $495.00.
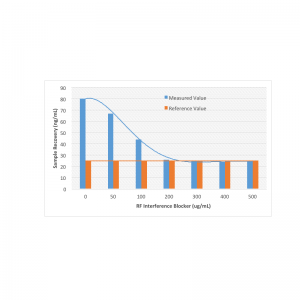
Rheumatoid Factor Interference Blocker
-
- Sale!
- Antibodies, Interference
Rheumatoid Factor Interference Blocker
- Original price was: $595.00.$395.00Current price is: $395.00.
New Antibodies from MOLECULAR DEPOT
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CTNL1 CTNNAL1 Antibody (Human, Mouse)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Monoclonal CXCL11 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal Cullin 4a (CUL4A) Antibody (Human)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal Cullin 1/CUL1 (N-Terminus) Antibody (Human, Mouse, Rat)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CTNA2 CTNNA2 Antibody (Human, Mouse)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Monoclonal CTR1/SLC31A1 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Mouse Monoclonal CTLA4 Antibody
- Original price was: $1,795.00.$895.00Current price is: $895.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CXCL5 (A38-Q130) Antibody (Rat)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CXCR1 (N-Terminus) Antibody (Human)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Anti-CXB5 GJB5 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
New Proteins from MOLECULAR DEPOT
-
- Sale!
- Conjugates, Proteins
Captopril HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Aminopenicillic Acid HRP Conjugate
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Biochemistry, Proteins
PKC-α, Active, GST-tagged from Xanopus sp.
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Conjugates, Proteins
Prilocain HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
T3 (Triiodothyronine) HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Pipemidic acid HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Biochemistry, Proteins
PKD2 Protein, Active (Recombinant Human)
- Original price was: $1,795.00.$895.00Current price is: $895.00.
-
- Sale!
- Conjugates, Proteins
Meropenem BSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Piperacillin HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Biochemistry, Proteins
Protein A Peroxidase from Staphylococcus aureus/horseradish
- Original price was: $1,195.00.$595.00Current price is: $595.00.
New Chemicals from MOLECULAR DEPOT
-
- Sale!
- Chemicals, Interference, Lipids
Triglyceride Mix for Interference Testing
- Original price was: $895.00.$395.00Current price is: $395.00.
-
- Sale!
- Chemicals
Microparticle Stabilizer Solution
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Enzyme Acceptor Stabilization Buffer
- Original price was: $495.00.$250.00Current price is: $250.00.
-
- Sale!
- Buffers & Solutions, Chemicals
Enzyme Donor Stabilization Buffer
- Original price was: $495.00.$250.00Current price is: $250.00.
-
- Sale!
- Chemicals, Conjugates
Progesterone Biotin Conjugate Solution
- Original price was: $650.00.$350.00Current price is: $350.00.
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Activation Buffer
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Washing Buffer
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Blocking Buffer
- Price range: $395.00 through $695.00
-
- Sale!
- Buffers & Solutions, Chemicals
Latex Microparticles Anti Aggregation Agent
- Price range: $295.00 through $3,500.00
-
- Sale!
- Chemicals
CPRG (Chlorophenol red-β-D-galactopyranoside)
- Original price was: $850.00.$395.00Current price is: $395.00.