Comprehensive genomic sequencing of paired ovarian cancers reveals discordance in genes that determine clinical trial eligibility.
Authors of this article are:
Fehniger JE, Berger AA, Juckett L, Elvin J, Levine DA, Zajchowski DA.
A summary of the article is shown below:
OBJECTIVE: We analyzed comprehensive genomic sequencing results from paired ovarian cancer samples to identify changes in mutational events over time.METHODS: DNA from paired FFPE tumor samples from 50 ovarian cancer patients in the Clearity Foundation Data Repository was analyzed for genomic mutations (GM), copy number alterations (CNA), microsatellite status (MS), tumor mutation burden (TMB), and loss of heterozygosity (LOH) by hybrid-capture, next-generation sequencing of up to 315 genes. Genomic profiles were compared between samples from the same patient. Poor quality results excluded 6 pairs from all analyses and 9 from CNA or LOH.RESULTS: Forty-four patients with predominantly advanced stage disease (34, 77%) and serous histology (31, 70%) received a median of 3 intervening treatment regimens (range 1-13). Analysis of 22 primary and recurrent sample pairs and 22 recurrent tumor pairs detected a median of 2 GM (range 0-5) and 1 CNA (range 0-6)/sample. TMB, MS, and LOH results were mostly concordant across paired samples. GM were consistent across most pairs [32/44 (73%) concordant], while CNA concordance was less [18/35 (51%)]. No changes were detected in therapeutically relevant GM, but 23% of patients had GM or CNA in the second sample that affect clinical trial eligibility.CONCLUSIONS: Paired ovarian cancer samples demonstrate stable genomic alterations across time. However, discordance was observed for some genes used as eligibility criteria for molecularly targeted clinical trials. Repeat tumor testing may be useful in cases where eligibility for such trials is deemed important after consideration of testing costs and potential clinical benefit.Copyright © 2019 Elsevier Inc. All rights reserved.
Check out the article’s website on Pubmed for more information:
[link-preview url=https://www.ncbi.nlm.nih.gov/pubmed/31703812 forceshot=true]
This article is a good source of information and a good way to become familiar with topics such as: Adult; Aged; Clinical Trials as Topic; DNA Mutational Analysis; DNA, Neoplasm; Female; Gene Dosage; High-Throughput Nucleotide Sequencing; Humans; Loss of Heterozygosity; Microsatellite Instability; Middle Aged; Neoplasm Staging; Ovarian Neoplasms.
NativeFolder: The Only Bacterial Culture Medium for the Expression of Soluble Proteins
-
- Sale!
- Molecular Biology, Research Kits
NativeFolder Bacterial Culture Medium
- Original price was: $795.00.$295.00Current price is: $295.00.
Interference Test Kit for Assay Validation
-
- Sale!
- Interference, Research Kits
Interference Test Kit
- Original price was: $995.00.$495.00Current price is: $495.00.
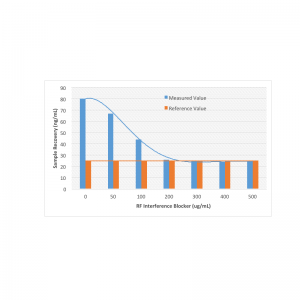
Rheumatoid Factor Interference Blocker
-
- Sale!
- Antibodies, Interference
Rheumatoid Factor Interference Blocker
- Original price was: $595.00.$395.00Current price is: $395.00.
New Antibodies from MOLECULAR DEPOT
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CTNL1 CTNNAL1 Antibody (Human, Mouse)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Monoclonal CXCL11 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal Cullin 4a (CUL4A) Antibody (Human)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal Cullin 1/CUL1 (N-Terminus) Antibody (Human, Mouse, Rat)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CTNA2 CTNNA2 Antibody (Human, Mouse)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Monoclonal CTR1/SLC31A1 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Mouse Monoclonal CTLA4 Antibody
- Original price was: $1,795.00.$895.00Current price is: $895.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CXCL5 (A38-Q130) Antibody (Rat)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Rabbit Polyclonal CXCR1 (N-Terminus) Antibody (Human)
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Antibodies, Biochemistry
Anti-CXB5 GJB5 Antibody
- Original price was: $1,595.00.$795.00Current price is: $795.00.
New Proteins from MOLECULAR DEPOT
-
- Sale!
- Conjugates, Proteins
Captopril HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Aminopenicillic Acid HRP Conjugate
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Biochemistry, Proteins
PKC-α, Active, GST-tagged from Xanopus sp.
- Original price was: $1,595.00.$795.00Current price is: $795.00.
-
- Sale!
- Conjugates, Proteins
Prilocain HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
T3 (Triiodothyronine) HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Pipemidic acid HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Biochemistry, Proteins
PKD2 Protein, Active (Recombinant Human)
- Original price was: $1,795.00.$895.00Current price is: $895.00.
-
- Sale!
- Conjugates, Proteins
Meropenem BSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Conjugates, Proteins
Piperacillin HSA Biotin Conjugate
- Original price was: $1,995.00.$995.00Current price is: $995.00.
-
- Sale!
- Biochemistry, Proteins
Protein A Peroxidase from Staphylococcus aureus/horseradish
- Original price was: $1,195.00.$595.00Current price is: $595.00.
New Chemicals from MOLECULAR DEPOT
-
- Sale!
- Chemicals, Interference, Lipids
Triglyceride Mix for Interference Testing
- Original price was: $895.00.$395.00Current price is: $395.00.
-
- Sale!
- Chemicals
Microparticle Stabilizer Solution
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Enzyme Acceptor Stabilization Buffer
- Original price was: $495.00.$250.00Current price is: $250.00.
-
- Sale!
- Buffers & Solutions, Chemicals
Enzyme Donor Stabilization Buffer
- Original price was: $495.00.$250.00Current price is: $250.00.
-
- Sale!
- Chemicals, Conjugates
Progesterone Biotin Conjugate Solution
- Original price was: $650.00.$350.00Current price is: $350.00.
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Activation Buffer
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Washing Buffer
- Price range: $295.00 through $495.00
-
- Sale!
- Buffers & Solutions, Chemicals
Microparticle Blocking Buffer
- Price range: $395.00 through $695.00
-
- Sale!
- Buffers & Solutions, Chemicals
Latex Microparticles Anti Aggregation Agent
- Price range: $295.00 through $3,500.00
-
- Sale!
- Chemicals
CPRG (Chlorophenol red-β-D-galactopyranoside)
- Original price was: $850.00.$395.00Current price is: $395.00.