 Expression of soluble proteins remains a major challenge for biochemists. Although E. coli remains the most straightforward, flexible, fast and inexpensive expression system of heterologous proteins, many proteins accumulate in the cytoplasm of the bacterium in an aggregated misfolded state known as inclusion bodies. Proteins in inclusion bodies are often inactive and attempt to denature and renature these inclusions bodies are time consuming, tedious and often ineffective as the renature proteins often fails to regain activity.
Expression of soluble proteins remains a major challenge for biochemists. Although E. coli remains the most straightforward, flexible, fast and inexpensive expression system of heterologous proteins, many proteins accumulate in the cytoplasm of the bacterium in an aggregated misfolded state known as inclusion bodies. Proteins in inclusion bodies are often inactive and attempt to denature and renature these inclusions bodies are time consuming, tedious and often ineffective as the renature proteins often fails to regain activity.
Rather than experimenting with the renaturation of inclusion bodies, efforts should focused on the expression of soluble proteins in E. coli. Several strategies have been developed over the years to improve the solubility of heterologous proteins in E. coli. These include:
- The use of weaker promoters
- The use of low copy number plasmids
- The co-expression of chaperone (such as GroES-GroEL and DnaK-DnaJ-GrpE)
- The use of specific E. coli strains such as (AD494 and Origami)
- Fusing the target protein to a soluble partner
- Lowering the expression temperature
- Using a modified culture medium
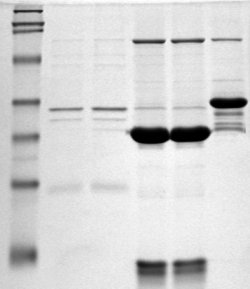
NativeFolder is bacterial growth medium specially formulated to help proteins fold correctly in E. coli and express as a soluble fraction. NativeFolder‘s pH, ionic strength, osmoprotectants and other additives promoted the soluble expression of several proteins (include certain membranes proteins) as large as 110 kDa.

